Global Point of Care
Determine™ HIV
Early Detect
FAST4WARD ACUTE HIV DETECTION
Determine™ HIV Early Detect is the first and only WHO pre-qualified 4th generation rapid diagnostic test (RDT).1
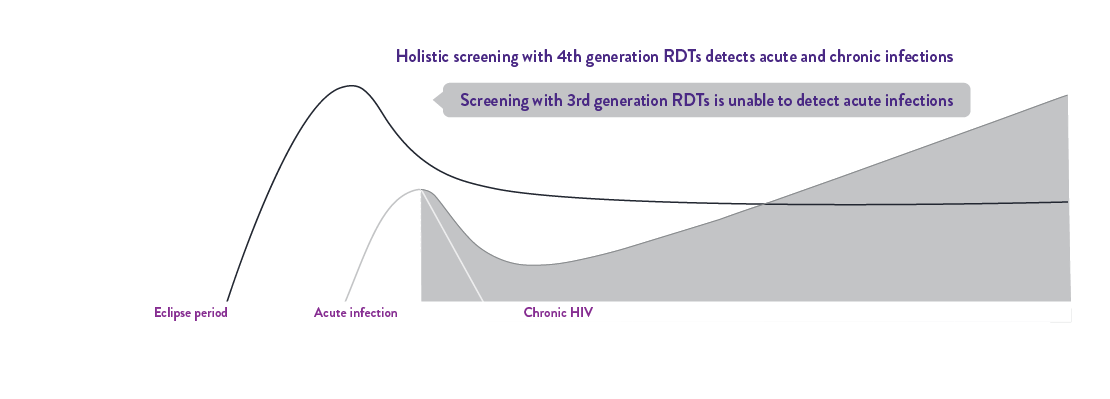
The 4th gen Determine™ HIV Early Detect sets a new standard in HIV point of care (POC) testing with its exceptional dual accuracy for holistic detection of acute and chronic HIV infections, protecting patients and communities.
Determine HIV Early Detect provides seamless testing and accurate results in just 20 minutes, using fingerstick or venipuncture whole blood, or serum/plasma.
Product exclusively available for export to select markets.

Benefits
EXCEPTIONAL DUAL ACCURACY
Accurately detects acute HIV infection with p24 antigen,2 and chronic HIV infection with its highest antibody analytical sensitivity,2 making it an ideal Assay 1 test.
MEANINGFUL CLINICAL AND ECONOMICAL IMPACT
Halts onward transmission and improves prognosis with significant clinical and economical value through acute HIV detection and treatment.3 Provides significant economic value by reducing costs associated with new transmissions and late diagnoses.4,5
SEAMLESS TESTING FOR IMMEDATE CARE
Simple test procedure that empowers user experience and ease of use through flexible testing workflow and extended reading time window.2
Determine HIV Early Detect has EXCEPTIONAL dual accuracy to detect both acute and chronic infections for a holistic screening approach.


Helpful Documents
PRODUCT DOCUMENTS
SPECIFICATIONS
- Sensitivity* 100% Specificity 99.72%
*Please refer to the Product Insert for the analytical sensitivity of HIV-1 p24 antigen - Method: Lateral Flow
- Time to results: 20 minutes
- Storage conditions: 2-30°C
- Sample volume: 50µl
- Test shelf life: 18 months
- Sample types: Serum/plasma, fingerstick whole blood and venipuncture whole blood
PRODUCT CODE
| Determine™ HIV Early Detect | CE-Marked | Non-CE Marked |
|---|---|---|
| Determine™ HIV Early Detect 20 Test Kit | 7D2842 | |
| Determine™ HIV Early Detect 100 Test Kit | 7D2843 | |
| Determine™ HIV Early Detect SET (100 tests and accessories)* | 7D2843SET | |
| Chase Buffer (for 100 tests) | 7D2243 | 7D2243 |
| EDTA Capillary Tubes (100) | 7D2222 |
* Investigational use only in Australia
VIDEOS

Venipuncture Whole Blood Test Procedure

Serum and Plasma Test Procedure
View References
- World Health Organization. Prequalified in vitro diagnostics. WHO Prequalification of Medical Products. Accessed August 6, 2025. https://extranet.who.int/prequal/vitro-diagnostics/prequalified/in-vitro-diagnostics
- WHO Prequalification of In Vitro Diagnostics. (2020). Public report: Product: Determine™ HIV Early Detect (PQDx-0243-013-00). World Health Organization. https://extranet.who.int/prequal/sites/default/files/whopr_files/PQDx_0243-013-00DetermineHIVEarlyDetect_v8.pdf
- Kroon EDMB, Phanuphak N, Shattock AJ, et al. Acute HIV infection detection and immediate treatment estimated to reduce transmission by 89% among men who have sex with men in Bangkok. J Int AIDS Soc. 2017;20(1):21708. doi:10.7448/IAS.20.1.21708
- Tran H, Saleem K, Lim M, et al. Global estimates for the lifetime cost of managing HIV. AIDS. 2021;35(8):1273-1281. https://doi.org/10.1097/qad.0000000000002887
- Fleishman JA, Yehia BR, Moore RD, Gebo KA; HIV Research Network. The economic burden of late entry into medical care for patients with HIV infection. Med Care. 2010;48(12):1071-1079. https://doi.org/10.1097/mlr.0b013e3181f81c4a
- Patel P, Bennett B, Sullivan T, Parker MM, Heffelfinger JD, Sullivan PS; CDC AHI Study Group. Rapid HIV screening: missed opportunities for HIV diagnosis and prevention. J Clin Virol. 2012 May;54(1):42-7. https://doi.org/10.1016/j.jcv.2012.01.022



