GLOBAL POINT OF CARE
OBJECTIEF
MILDE TBI BEOORDELEN
OP HET POINT-OF-CARE
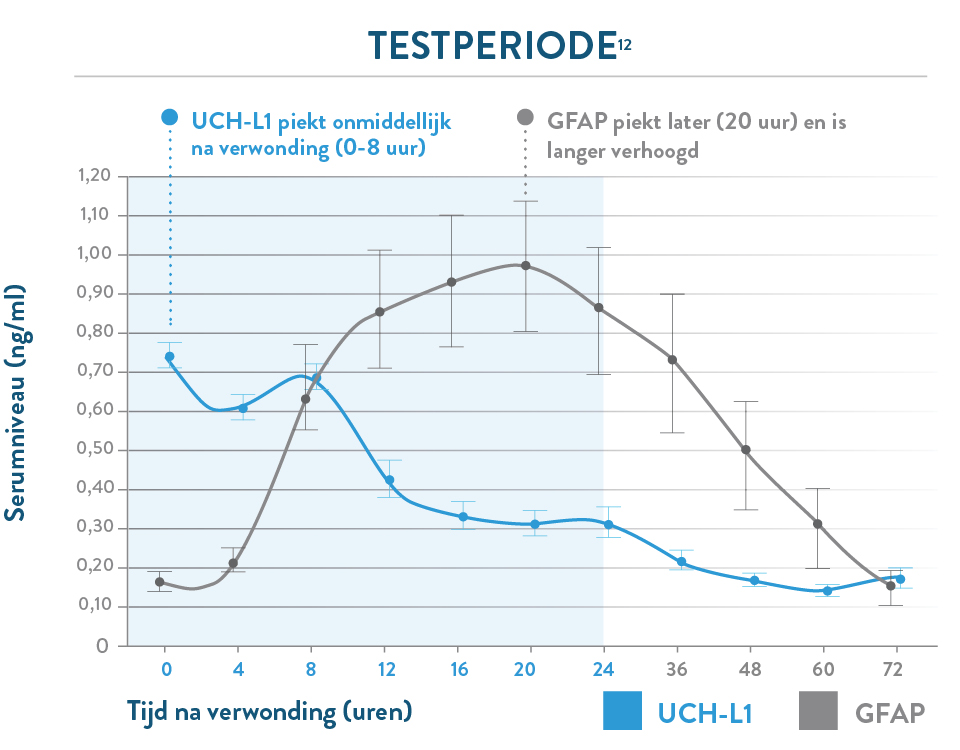
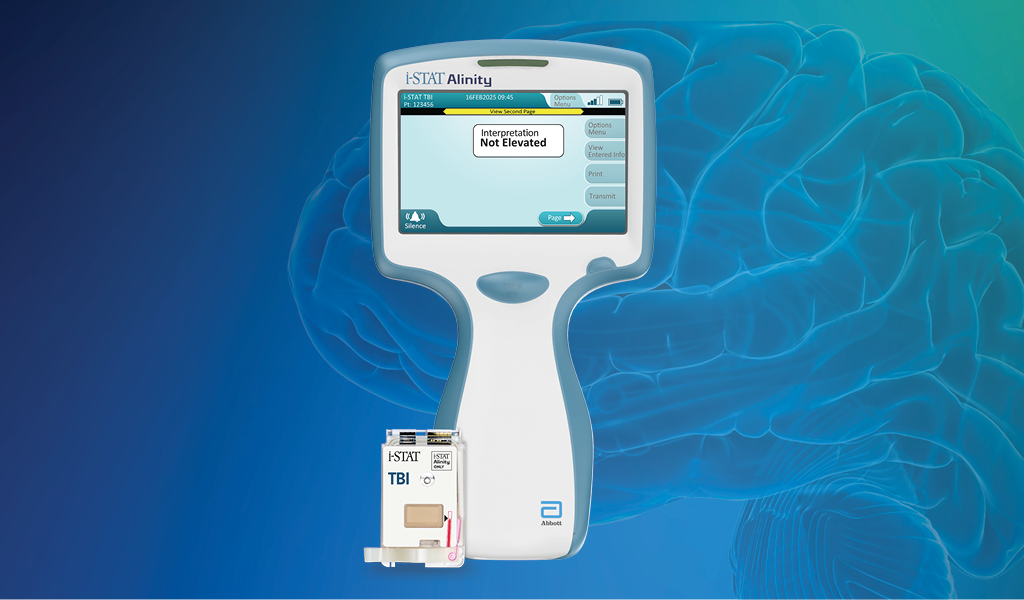
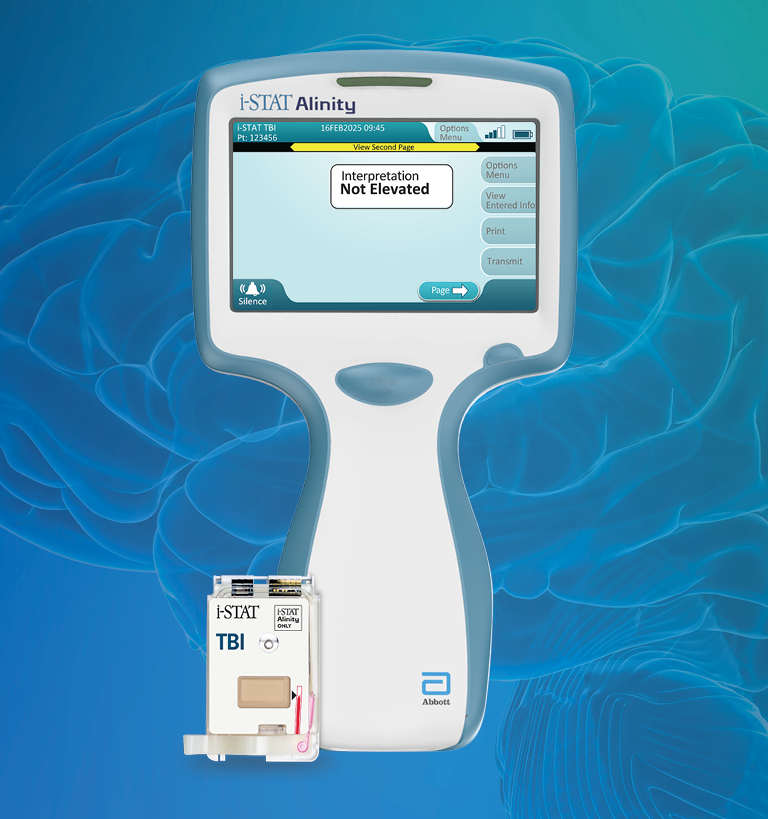
De i-STAT TBI cartridge* is de eerste point-of-care veneuze volbloedtest voor milde TBI die hersenspecifieke biomarkers meet en objectieve gegevens levert in 15 minuten.1
*Alleen beschikbaar op het i-STAT Alinity-instrument | TBI=traumatisch hersenletsel
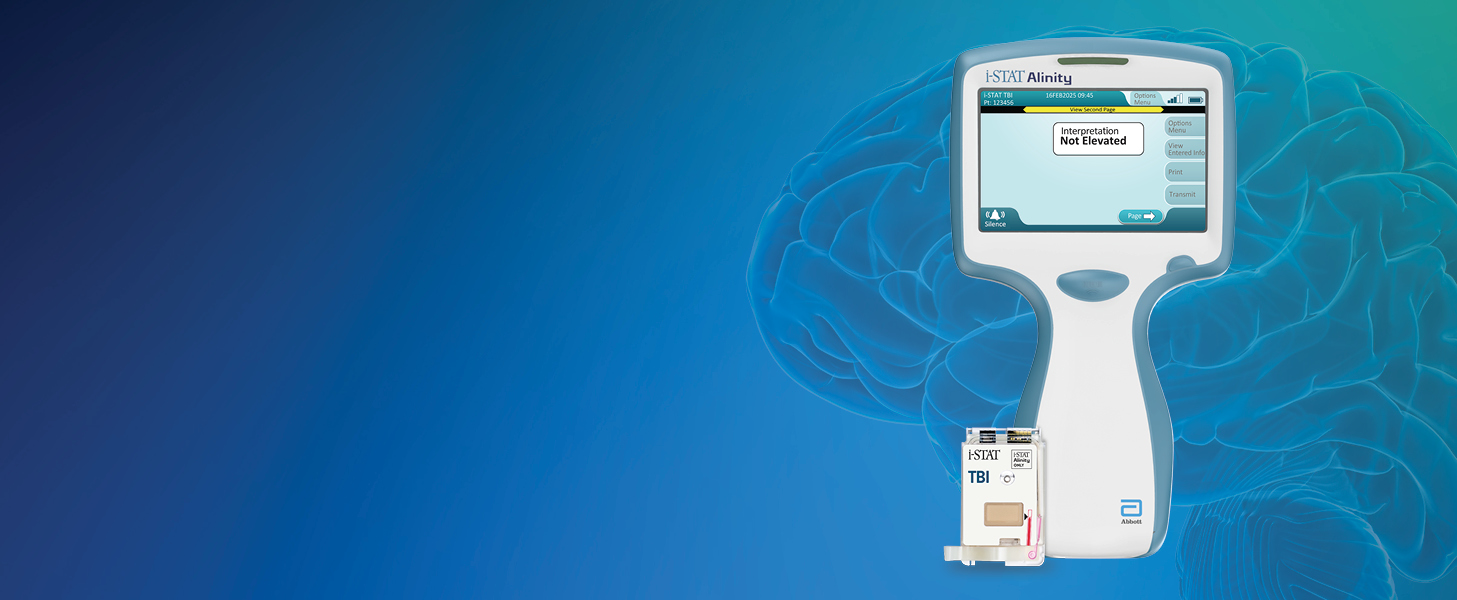

OBJECTIEF
MILDE TBI BEOORDELEN
OP HET POINT-OF-CARE
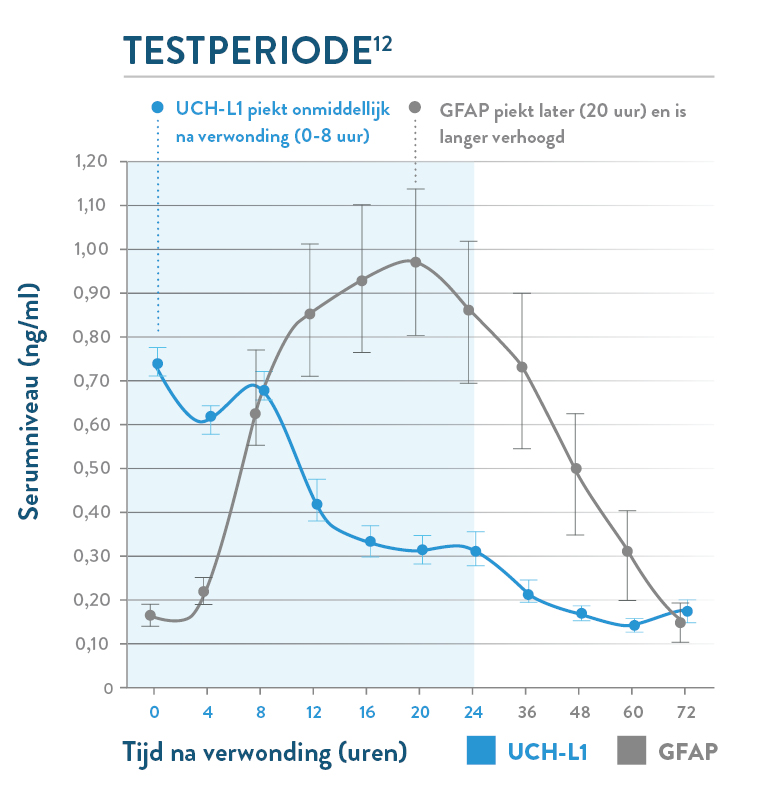
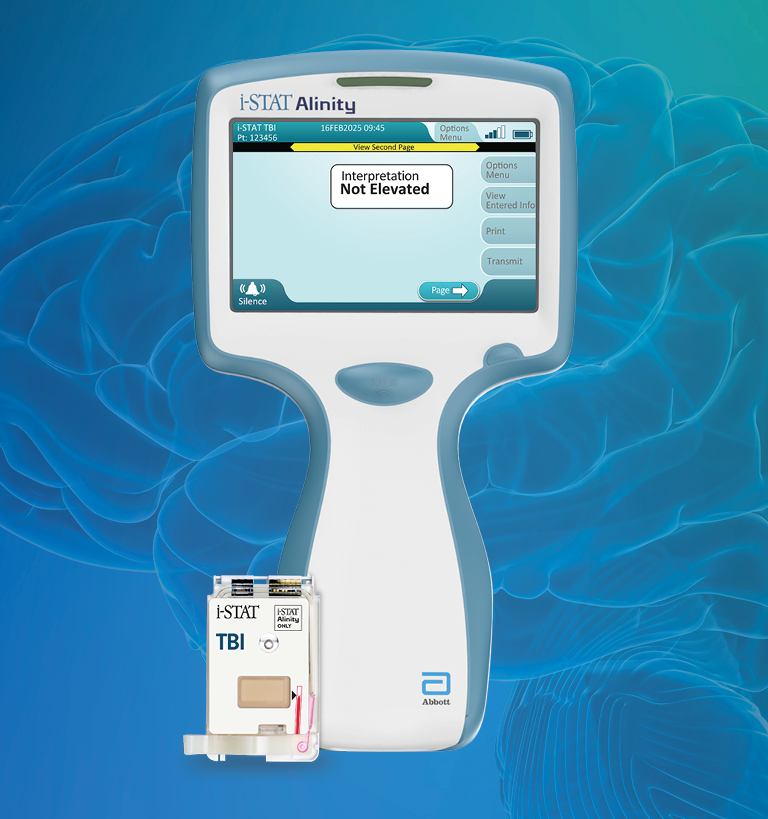
De i-STAT TBI cartridge* is de eerste point-of-care veneuze volbloedtest voor milde TBI die hersenspecifieke biomarkers meet en objectieve gegevens levert in 15 minuten.1
*Alleen beschikbaar op het i-STAT Alinity-instrument | TBI=traumatisch hersenletsel
INNOVATIES IN PATIËNTENZORG
Welke impact zal de i-STAT TBI cartridge hebben op uw patiënten en zorgteam? Neem contact met ons op en ontdek onze innovatieve point-of-care producten die binnen enkele minuten laboratoriumresultaten leveren.




INNOVATIES IN PATIËNTENZORG
Welke impact zal de i-STAT TBI-cartridge hebben op uw patiënten
en zorgteam?
AANVULLENDE BRONNEN

In deze whitepaper wordt meer uitgelegd over de rol die biomarkers spelen bij de beoordeling van mild traumatisch hersenletsel (in het Engels).

Ontdek meer over de rol van biomarkers (GFAP en UCH-L1) bij het beoordelen van mild traumatisch hersenletsel (mTBI) (in het Engels).
Referenties
- i-STAT TBI cartridge. Instructions for use. Abbott Point of Care Inc. Abbott Park, IL; 2024.
- Zetterberg H, Blennow K. Fluid biomarkers for mild traumatic brain injury and related conditions. Nat Rev Neurol. 2016;12(10):563-574.
- Chodobski A, Zink BJ, Symydynger-Chodobska J. Blood-brain barrier pathophysiology in traumatic brain injury. Transl Stroke Res. 2011;2(4):492-516.
- Metting Z, Wilczak N, Rodiger LA, et al. GFAP and S100B in the acute phase of mild traumatic brain injury. Neurology. 2012;78(18):1428-1433.
- Papa L, Lewis LM, Falk JL, et al. Elevated levels of serum glial fibrillary acidic protein breakdown products in mild and moderate traumatic brain injury are associated with intracranial lesions and neurosurgical intervention. Ann Emerg Med. 2012;59(6):471-483.
- Jones A, Jarvis P. Review of the potential use of blood neuro-biomarkers in the diagnosis of mild traumatic brain injury. Clin Exp Emerg Med. 2017;4(3):121-127.
- Schulte S, Podlog LW, Hamson-Utley JJ, et al. A systematic review of the biomarker S100B: implications for sport-related concussion management. J Athl Train. 2014;49(6):830-850.
- Steiner J, Bernstein H-G, Bielau H, et al. Evidence for a wide extra-astrocytic distribution of S100B in human brain. BMC Neurosci. 2007;8:2.
- Pelinka LE, Kroepfl A, Schmidhammer R, et al. Glial fibrillary acidic protein in serum after traumatic brain injury and multiple trauma. J Trauma. 2004;57(5):1006-1012.
- Diaz-Arrastia R, Wang KKW, Papa L, et al. Acute biomarkers of traumatic brain injury: relationship between plasma levels of ubiquitin C-terminal hydrolase-L1 and glial fibrillary acidic protein. J Neurotrauma. 2014;31(1):19-25.
- Papa L, Lewis LM, Silvestri S, et al. Serum levels of ubiquitin C-terminal hydrolase (UCH-L1) distinguish mild traumatic brain injury (TBI) from trauma controls and are elevated in mild and moderate TBI patients with intracranial lesions and neurosurgical intervention. J Trauma Acute Care Surg. 2012;72(5):1335-1344.
- Papa L, Brophy GM, Welch RD, et al. Time course and diagnostic accuracy of glial and neuronal blood biomarkers GFAP and UCH-L1 in a large cohort of trauma patients with and without mild traumatic brain injury. JAMA Neurol. 2016;73(5):551-560.