Global Point Of Care
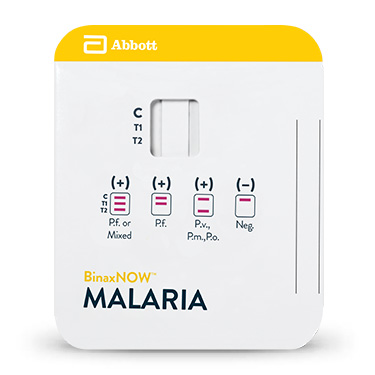
BinaxNOW™ Malaria
O teste BinaxNOW™ Malária é um teste imunocromatográfico in vitro para a detecção qualitativa de antígenos de Plasmodium que circulam no sangue de pessoas com sinais e sintomas de infecção por malária. O teste está voltado para detecção do antígeno da proteína rica em histidina II (HRPII) específica de Plasmodium falciparum (P.f.) e um antígeno pan-malário comum a todas as quatro espécies de malária capazes de infectar humanos com
P. falciparum, P. vivax (P.v.), P. ovale (P.o.) e P. malariae (P.m.).
Destina-se a auxiliar no rápido diagnóstico de infecções de malária humana e no diagnóstico diferencial de infecções por Plasmodium falciparum (P.f.) de outras infecções malárias menos virulentas. Resultados negativos devem ser confirmados por microscopia de esfregaço espesso/fino.