Global Point of Care
Determine™ HIV-1/2 Ag/Ab Combo
The first FDA approved rapid point-of-care test that detects both HIV-1/2 antibodies and free HIV-1 p24 antigen. This antigen/antibody test (4th generation) has the ability to identify HIV earlier than antibody-only tests (2nd and 3rd generation).1 It enables health care providers to diagnose HIV infection earlier allowing individuals to seek medical care sooner.
Only available in the US.

Benefits
CLIA-waived for fingerstick whole blood
Innovative
It’s the first antigen/antibody rapid point-of-care test that detects both HIV-1/2 antibodies and free HIV-1 p24 antigen on a single test strip.
Dependable
Built-in quality controls to let you know the test is working.
Efficient
Test in three simple steps with clear results in just 20 minutes.
Earlier Detection
The first antigen/antibody test, with the ability to detect HIV earlier than antibody-only tests.1
Flexible
Test using whole blood, serum or plasma samples.
Reliable
Proved 99.9% overall clinical sensitivity for all sample types.
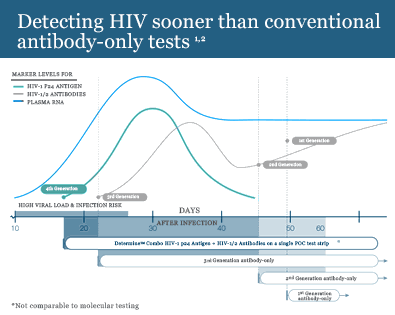


Click image for full size view.
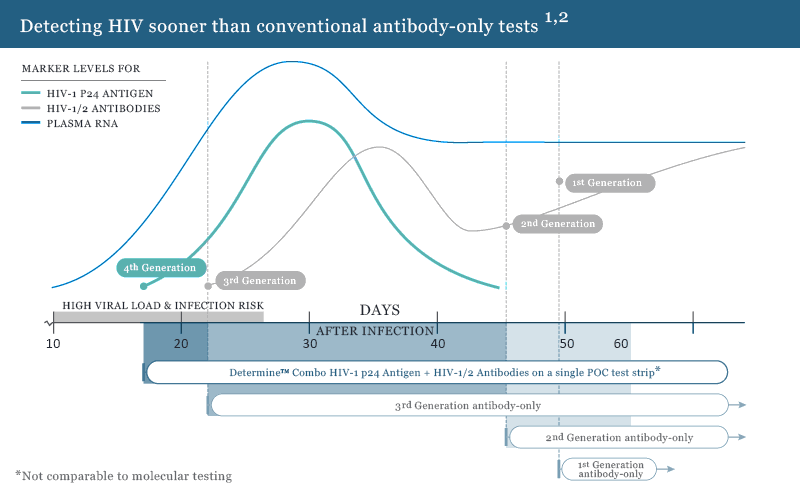
By detecting HIV earlier, you are helping to prevent further transmission during this extremely infectious early stage.



Helpful Documents
PRODUCT DOCUMENTS
SPECIFICATIONS
Information Type | Product Detail |
Method | Lateral Flow |
Time to results | 20 minutes |
Results window | 20-30 minutes after starting test |
Test lines | HIV-1 p24 antigen |
Storage conditions | 2-30°C (36-86°F) |
Test shelf life | 24 months* |
External controls shelf life | 24 months** |
Sample type | Wholeblood/serum/plasma |
Operating temperature | 15-30°C (59 to 86°F) |
Sample Type | Overall Clinical | Overall Clinical |
Fingerstick Whole Blood | 99.9% | 99.8% |
Venous Whole Blood | 99.9% | 99.7% |
Serum | 99.9% | 99.6% |
Plasma | 99.9% | 99.7% |
PRODUCT CODE
Determine™ HIV-1/2 Ag/Ab | Code
|
Determine™ HIV-1/2Ag/Ab Combo (25 Tests) | 7D2648 |
External Controls | 7D2628 |
Verification Panel | KZMC030 |
VIDEOS
Determine Venipuncture Samples Test Procedure Demonstration
Buy Now
Order your test kits online from our authorized distributors or contact an Abbott sales representative.
View References
1. Masciotra, S, et al. Performance of the Alere Determine™ HIV-1/2 Ag/Ab Combo Rapid Test with specimens from HIV-1 seroconverters from the US and HIV-2 infected individuals from Ivory Coast. J Clin Virol 2013: Published online 05 August 2013. DOI: 10.1016/j.jcv.2013.07.002 http://www.journalofclinicalvirology.com/article/S1386-6532(13)00277-1/abstract
2. Fiebig EW, Wright DJ, Rawal BD, et al. Dynamics of HIV viremia and antibodyseroconversion in plasma donors: implications for diagnosis and staging of primary HIV infection. AIDS. 2003;17(13):1871-1879.
*Average shelf life received by customer (product has a 15 month shelf life from date of manufacture)
**From date of manufacture